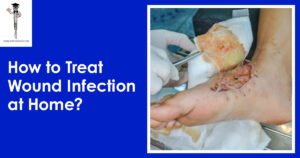
How to Treat Wound Infection at Home?
Dealing with a wound infection at home can be daunting, but it can be done correctly. Taking good care of wounds is an essential part of wound management to avoid complications and

Dealing with a wound infection at home can be daunting, but it can be done correctly. Taking good care of wounds is an essential part of wound management to avoid complications and
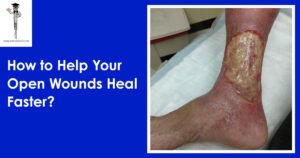
Imagine healing from open wounds more quickly, easily, and without ever leaving the comfort of your home; sounds ideal, right? Open wounds need immediate, professional treatment to reduce the chance

When it comes to wound care, time is not just money- it is life! Delay in taking care of a wound, or not, can make the difference between life and

Daniel Davidson, MD, MBA, DBA, PHD Introduction: A common surgical technique called skin grafting involves transferring healthy skin from one area of the body to another in order to cure

Daniel Davidson, MD, MBA, DBA, PHD Introduction: Debridement of the wound entails removing diseased, damaged, or dead tissue from the wound bed; it is an essential part of wound care.

Daniel Davidson, MD, MBA, DBA, PHD Introduction: A common wound care method is called “wet-to-dry dressing,” which entails moistening a wound and letting it dry before removing it. This technique